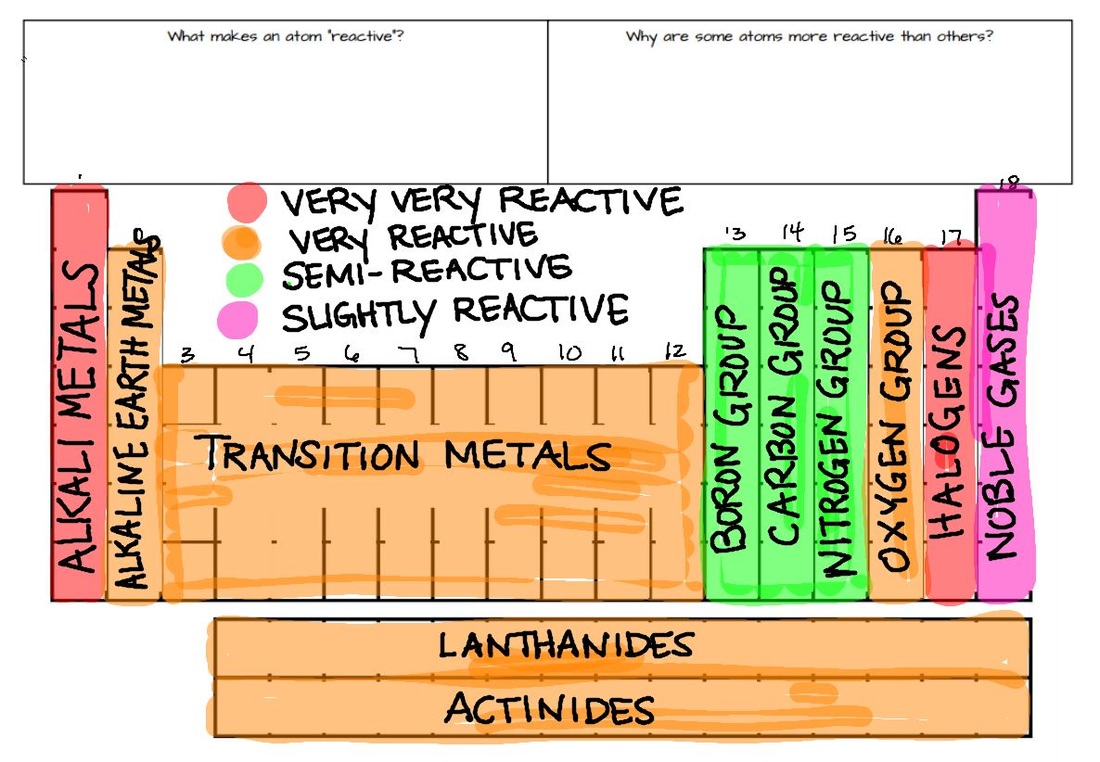
Metals and non-metals in the periodic table They have different electronic structure, and this influences the way in which they react. Trends Across the Period: The non-metallic character of the chemical elements increases as we move from the left to right along the period in a periodic table.Metals and non-metals are found in different parts of the periodic table.Trends Across the Group: The non-metallic character of the chemical elements decreases as we move from the top to bottom in a group in the periodic table.Trend Across the Group: The metallic character of the chemical elements increases as we move from the top to bottom in a group in the periodic table.Trend Across the Period: The metallic character of the chemical elements decreases as we move from the left to right along the period in a periodic table.Reason: Electron-loving character increases with the increase in nuclear charge. Trend Across the Period: The electronegativity increases as we move from left to right in a period.Trend Across the Group: The electronegativity decreases as we move from top to bottom in a period.Reason: Increase in nuclear charge and decrease in atomic radius.ĮLECTRONEGATIVITY: The ability of an atom to attract paired electrons toward itself for the formation of a chemical bond with another atom. Trend Across the Period: The ionization energy increases as we move from left to right in a row horizontally.Reason: Increase in atomic radius across the group. Trend Across the Group: The ionization energy decreases as we move from top to bottom in a group in the periodic table.IONIZATION ENERGY: The amount of energy that is required to remove the outermost electron of the atom is called ionization energy. Reason: Nuclear charge increases but atomic radius decreases. Trend Across the Period: As we move from left to right in a period, the electron affinity increases.Trend Across the Group: As we move from top to bottom in a group, electron affinity decreases.Reason: Nuclear charge increases but the number of shells remains the same.ĮLECTRON AFFINITY: The amount of energy required to add an electron to an atom is called electron affinity. Trend Across the Period: As we move from left to right in a period, the atomic radius decreases gradually.Reason: There are two reasons for the increase in atomic radius across the group that is increasing the number of shells and an increase in nuclear charge. 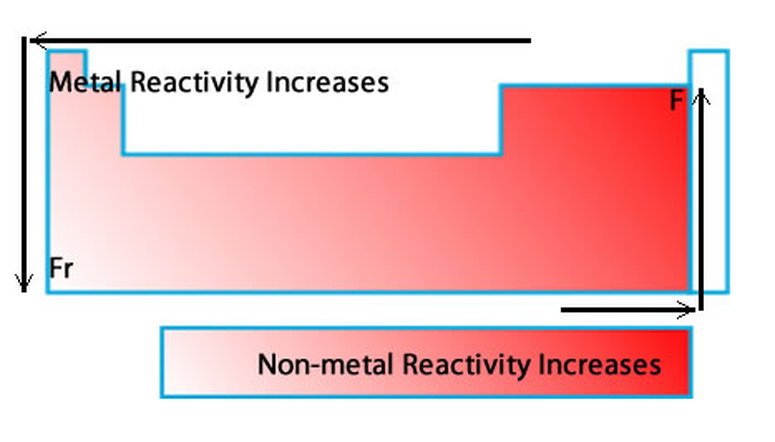
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
